"...a defining tenet for catalyst generation has been the mimicry and incorporation of a reaction's stereoelectronics into hapten design. Success necessarily depends upon accurate assumptions of mechanistic details, the extent to which the hapten is a model of the transition state, and how the immune system responds to the antigen."
Kim Janda, J. Am. Chem. Soc. 1998, 120, 2212
Each of these issues can be addressed with modern computational chemistry. First, mechanistic details for the reactions whose catalysis is desired can be obtained by quantum mechanical methods. The structures of transition states are computed, allowing for a direct assessment of the similarities and differences between them and the haptens designed to mimic them. In addition, bioinformatics and computational docking studies can be used to garner insight into the processes by which the immune system produces effective catalysts. Information from such studies can then be used to improve catalysis by suggesting alterations to haptens and known antibodies.
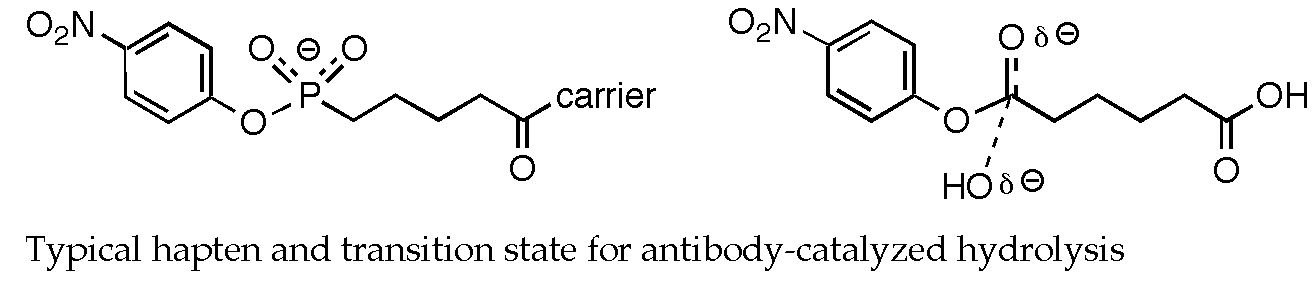
To test this strategy for elucidating the origins of antibody catalysis, the hydrolyses of phenyl and para-nitrophenyl acetate were examined in the gas phase, in solution, and in the combining sites of various antibody catalysts. Intermediates and transition states involved in gas phase hydrolysis were investigated using Hartree-Fock calculations. The structures of these species were then compared with those of phosphonate transition state analogs used as haptens to elicit hydrolytic antibodies. Although many similarities were observed, several important differences were noted. In particular, the asymmetry of the transition state for hydroxide addition (the rate-determining step) is not faithfully reproduced in existing haptens, a situation that can potentially be remedied through hapten redesign.
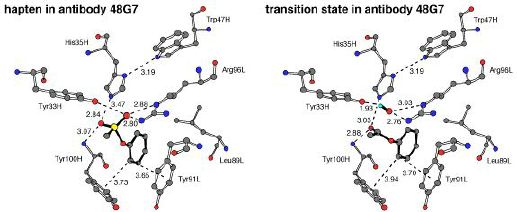
Computed transition states were then docked into the crystallographically-determined structures of various hydrolytic antibodies using a Monte Carlo simulated annealing technique. These studies showed that, although haptens and transition states do bind to hydrolytic antibodies in similar orientations, optimal stabilization of each involves differently arrayed hydrogen bond donor and acceptor groups (see figure below). Computational mutants were designed based on these observations, and ultimately we hope to test these predictions experimentally.
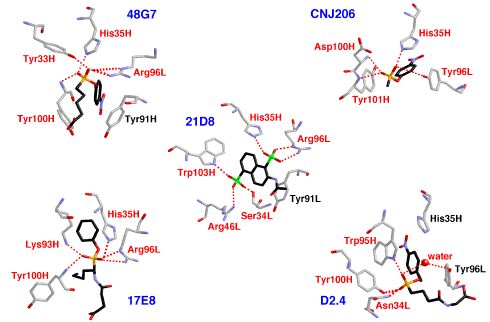
In addition, comparison of known antibody structures and sequences using bioinformatic strategies has provided insights into the nature of the immune response that leads to catalysts. For example, such experiments suggested the presence of generic binding sites present in the naive repertoire of antibodies that may be capable of binding phosphonate haptens, hydrolytic transition states, arsonate haptens, sulfonate haptens, and nucleotides - all molecules that contain tetrahedral anionic substructures (see figure below). Principles such as this govern the production of mature antibodies and can be exploited by chemists attempting to engineer efficient antibody catalysts.
Tantillo, D. J.; Houk, K. N. J. Org. Chem. 1999, 64, 3066-3076: "Fidelity in Hapten Design: How Analogous Are Phosphonate Haptens to the Transition States for Alkaline Hydrolyses of Aryl Esters?"
Hotta, K.; Lange, H.; Tantillo, D. J.; Houk, K. N.; Hilvert, D.; Wilson, I. A. J. Mol. Biol. 2000, 302, 1213-1225: "Catalysis of Decarboxylation by a Preorganized Heterogenous Microenvironment: Crystal Structures of Abzyme 21D8"
Tantillo, D. J.; Houk, K. N. Chem. Biol. 2001, 8, 535-545: "Canonical Binding Arrays as Molecular Recognition Elements in the Immune System: Tetrahedral Anions and the Ester Hydrolysis Transition State"
Tantillo, D. J.; Houk, K. N. J. Comp. Chem. 2002, 23, 84-95.: "Transition State Docking: A Probe for Noncovalent Catalysis in Biological Systems. Application to Antibody-Catalyzed Ester Hydrolysis," part of a special issue on biological aspects of computational chemistry.
Ujaque, G.; Tantillo, D. J.; Hu, Y.; Houk, K. N.; Hotta, K.; Hilvert, D. J. Comp. Chem. 2002, 24, 98-110: "Catalysis on the Coastline: Theozyme, Molecular Dynamics, and Free Energy Perturbation Analysis of Antibody 21D8 Catalysis of the Decarboxylation of 5-Nitro-3-Carboxybenzisoxazole," part of a special issue honoring Dr. Peter A. Kollman.
Tantillo, D. J.; Leach, A.; Zhang, X.; Houk, K. N: "Theoretical Studies of Antibody Catalysis" In Catalytic Antibodies, Wiley-VCH: Weinheim, Germany, 2004, pp. 72-117.
Houk, K. N.; Tantillo, D. J. CHEMTRACTS: Organic Chemistry 1997, 10, 1039-1041: Condensation and commentary on "Relaxing Substrate Specificity in Antibody-Catalyzed Reactions: Enantioselective Hydrolysis of N-Cbz-Amino Acid Esters - Research by Tanaka, F.; Kinoshita, K.; Tanimura, R.; Fujii, I."
Dean J. Tantillo and K. N. Houk: "Stabilization of Transition States and Their Analogs by Hydrolytic Antibodies" Poster presented at the 216th ACS National Meeting, Boston, MA, August 23-27, 1998; paper ORGN 649.
Dean J. Tantillo and K. N. Houk: "Analogies Between Antibody Hydrolases and Decarboxylases" Poster presented at the 5th Annual Maria Goeppert-Mayer Interdisciplinary Symposium, San Diego, CA, March 4, 2000
Dean J. Tantillo, Kinya Hotta, Donald Hilvert, and K. N. Houk: "Origins of Catalysis and Cross-Reactivity for an Antibody Decarboxylase" Poster presented at the 219th ACS National Meeting, San Francisco, CA, March 26-30, 2000; paper ORGN 422.
Dean J. Tantillo: "Theoretical Bioorganic Chemistry of Antibody Catalysis: Understanding an Antibody Decarboxylase" Lecture presented at the UCLA Department of Chemistry and Biochemistry Organic Chemistry Graduate Symposium, Honoring Christopher S. Foote on His 65th Birthday, Los Angeles, CA, June 3, 2000
back to list of research projects